Aniruddha Belsare
Affiliations Personal homepage Professional homepagehttp://cfwe.auburn.edu/profile/aniruddha-belsare/
ORCID more infohttps://orcid.org/0000-0002-4651-0116
GitHub more info
Aniruddha Belsare is a disease ecologist with a background in veterinary medicine, interspecific transmission, pathogen modeling and conservation research. Aniruddha received his Ph.D. in Wildlife Science (Focus: Disease Ecology) from the University of Missouri in 2013 and subsequently completed a postdoctoral fellowship there (University of Missouri, May 2014 – June 2017). He then was a postdoctoral fellow in the Center for Modeling Complex Interactions at the University of Idaho (June 2017 - March 2019) and later a Research Associate with the Boone and Crockett Quantitative Wildlife Center, Michigan State University (March 2019 - Jan 2021). He was a Research Scientist in the Civitello Disease Ecology Lab at Emory University from Jan 2021 to Jan 2023. Currently, Aniruddha is an Assistant Professor of Disease Ecology at the College of Forestry, Wildlife & Environment / College of Veterinary Medicine at Auburn University.
Research Interests
My research interests primarily lie at the interface of ecology and epidemiology, and include host-pathogen systems that are of public health or conservation concern. I use ecologic, epidemiologic and model-based investigations to understand how pathogens spread through, persist in, and impact host populations. Animal disease systems that I am currently working on include canine rabies, leptospirosis, chronic wasting disease, bighorn sheep pneumonia, raccoon roundworm (Baylisascaris procyonis), chytridiomycosis, and Lyme disease.
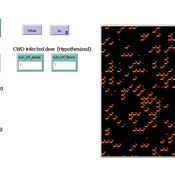
Peer reviewed CapOvCWD
Aniruddha Belsare | Published Tuesday, September 09, 2025 | Last modified Tuesday, November 11, 2025CapOvCWD is an agent-based model that simulates a captive cervid herd composed of adults and fawns. The model deer population is initialized using data on herd size and composition from captive facility records. Individual deer domiciliary history and annual CWD testing records inform the herd size and sample size (for CWD testing), respectively. The model can be used to iteratively estimate the facility level annual CWD detection probability. Detection probability estimates can be further refined by incorporating multiyear CWD testing data. This approach can be particularly useful for interpreting negative test results from a subset of the captive herd. Facility level detection probability estimates provide a comprehensive and standardized risk metric that reflects the likelihood of undetected CWD in the facility.
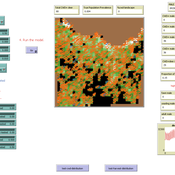
Peer reviewed MOOvPOP
Matthew Gompper Aniruddha Belsare Joshua J Millspaugh | Published Monday, April 10, 2017 | Last modified Saturday, April 19, 2025MOOvPOP is designed to simulate population dynamics (abundance, sex-age composition and distribution in the landscape) of white-tailed deer (Odocoileus virginianus) for a selected sampling region.
Peer reviewed MOOvPOPsurveillance
Matthew Gompper Aniruddha Belsare Joshua J Millspaugh | Published Tuesday, April 04, 2017 | Last modified Tuesday, May 12, 2020MOOvPOPsurveillance was developed as a tool for wildlife agencies to guide collection and analysis of disease surveillance data that relies on non-probabilistic methods like harvest-based sampling.
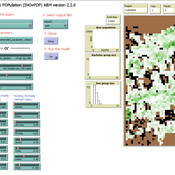
Peer reviewed INOvPOP
Aniruddha Belsare | Published Wednesday, June 01, 2022 | Last modified Wednesday, July 10, 2024INOvPOP is designed to simulate population dynamics (abundance, sex-age composition and distribution in the landscape) of white-tailed deer (Odocoileus virginianus) for selected Indiana counties. Updated for netLogo 6.4.0
Peer reviewed INOvCWD
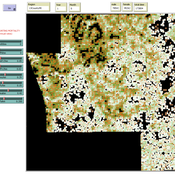
Aniruddha Belsare | Published Wednesday, June 01, 2022 | Last modified Wednesday, July 10, 2024INOvCWD is a spatially-explicit, agent-based model designed to simulate the spread of chronic wasting disease (CWD) in Indiana’s white-tailed deer populations.
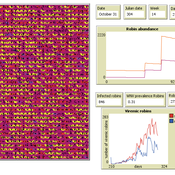
Peer reviewed AMRO_CULEX_WNV
Aniruddha Belsare Jennifer Owen | Published Saturday, February 27, 2021 | Last modified Thursday, March 11, 2021An agent-based model simulating West Nile Virus dynamics in a one host (American robin)-one vector (Culex spp. mosquito) system. ODD improved and code cleaned.
Peer reviewed DogPopDy: ABM for ABC planning
Aniruddha Belsare Abi Vanak | Published Saturday, August 01, 2020An agent-based model designed as a tool to assess and plan free-ranging dog population management programs that implement Animal Birth Control (ABC). The time, effort, financial resources and conditions needed to successfully control dog populations and achieve rabies control can be determined by performing virtual experiments using DogPopDy.
Peer reviewed MIOvPOPsurveillance
Aniruddha Belsare | Published Monday, April 13, 2020MIOvPOPsurveillance is set up to simulate harvest-based chronic wasting disease (CWD) surveillance of white-tailed deer (Odocoileus virginianus) populations in select Michigan Counties. New regions can be readily added, also the model can be readily adapted for other disease systems and used for informed-decision making during planning and implementation stages of disease surveillance in wildlife and free-ranging species.
Peer reviewed MIOvPOP
Aniruddha Belsare | Published Wednesday, September 18, 2019An ABM simulating white-tailed deer population dynamics for selected Michigan counties. The model yields pre-harvest and post-harvest realistic population snapshots that can be used to initialize the surveillance model (MIOvPOPsurveillance) and the CWD transmission dynamics model (MIOvCWD) respectively.
Peer reviewed BHSPopDy (Bighorn sheep population dynamics)
Aniruddha Belsare Ryan Long E Frances Cassirer | Published Tuesday, November 05, 2019This is an agent-based model coded in NetLogo. The model simulates population dynamics of bighorn sheep population in the Hell’s Canyon region of Idaho and will be used to develop a better understanding of pneumonia dynamics in bighorn sheep populations. The overarching objective is to provide a decision-making context for effective management of pneumonia in wild populations of bighorn sheep.
Peer reviewed DogFoxCDVspillover
Aniruddha Belsare Matthew Gompper | Published Thursday, March 16, 2017 | Last modified Tuesday, April 04, 2017The purpose of this model is to better understand the dynamics of a multihost pathogen in two host system comprising of high densities of domestic hosts and sympatric wildlife hosts susceptible to the pathogen.
Peer reviewed MIOvCWD
Aniruddha Belsare | Published Friday, December 13, 2019MIOvCWD is a spatially-explicit, agent-based model designed to simulate the spread of chronic wasting disease (CWD) in Michigan’s white-tailed deer populations. CWD is an emerging prion disease of North American cervids (white-tailed deer Odocoileus virginianus, mule deer Odocoileus hemionus, and elk Cervus elaphus) that is being actively managed by wildlife agencies in most states and provinces in North America, including Michigan. MIOvCWD incorporates features like deer population structure, social organization and behavior that are particularly useful to simulate CWD dynamics in regional deer populations.
Peer reviewed MHMSLeptoDy (Multi-host, multi-serovar Leptospira Dynamics Model)
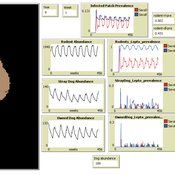
Aniruddha Belsare Matthew Gompper Meghan Mason Claudia Munoz-Zanzi | Published Tuesday, January 29, 2019 | Last modified Tuesday, March 12, 2019Leptospirosis is a neglected, bacterial zoonosis with worldwide distribution, primarily a disease of poverty. More than 200 pathogenic serovars of Leptospira bacteria exist, and a variety of species may act as reservoirs for these serovars. Human infection is the result of direct or indirect contact with Leptospira bacteria in the urine of infected animal hosts, primarily livestock, dogs, and rodents. There is increasing evidence that dogs and dog-adapted serovar Canicola play an important role in the burden of leptospirosis in humans in marginalized urban communities. What is needed is a more thorough understanding of the transmission dynamics of Leptospira in these marginalized urban communities, specifically the relative importance of dogs and rodents in the transmission of Leptospira to humans. This understanding will be vital for identifying meaningful intervention strategies.
One of the main objectives of MHMSLeptoDy is to elucidate transmission dynamics of host-adapted Leptospira strains in multi-host system. The model can also be used to evaluate alternate interventions aimed at reducing human infection risk in small-scale communities like urban slums.
Under development.